EXPERT VIEW/REVIEW PAPER |
https://doi.org/10.5005/jp-journals-10040-1236 |
Lisfranc Fracture Dislocation: A Review
1-3Department of Orthopedics Unit 1, Christian Medical College Vellore, Tamil Nadu, India
Corresponding Author: Manasseh Nithyananth, Department of Orthopedics Unit 1, Christian Medical College Vellore, Tamil Nadu, India, Phone: +91 4162282020, e-mail: manasseh@cmcvellore.ac.in
Received on: 28 February 2022; Accepted on: 28 March 2022; Published on: 31 December 2022
ABSTRACT
Introduction: Lisfranc joint is the articulation between midfoot and forefoot which is composed of the five tarsometatarsal joints, named after Jacques Lisfranc de Saint-Martin who was a field surgeon in Napoleon’s army.
Epidemiology and background: Lisfranc injuries constitute approximately 0.2% of all fractures. Although rare, they can be missed by the treating orthopedic surgeon, since present literature knowledge reveals that 20% of these injuries are missed or diagnosed late. As the consequences of missing such injuries are debilitating for the patients who end up with serious disability.
Diagnosis: To make accurate diagnosis, strong background knowledge of the anatomy of the Lisfranc joint is mandatory. Plain and stress radiographs, computed tomography (CT) scans, and magnetic resonance imaging (MRI) are augments to proper history taking and clinical examination and help in diagnosis especially in subtle unstable injuries.
Treatment: Stable injuries are treated nonoperatively. Unstable injuries are treated surgically; here, anatomical reduction and stable fixation is the standard of care. If closed reduction is unsatisfactory, open reduction should be performed. There are many modalities of fixation available, however, final outcome depends on early diagnosis, achieving anatomic reduction and maintaining it, irrespective of the method of reduction or the mode of fixation.
Complications: The common complications include osteoarthritis, persistent pain and gait abnormalities, implant-related problems, and skin problems.
How to cite this article: Bliss J, Albert S, Nithyananth M. Lisfranc Fracture Dislocation: A Review. J Foot Ankle Surg (Asia-Pacific) 2023;10(1):38-45.
Source of support: Nil
Conflict of interest: None
Keywords: Diagnosis, Fixation, Lisfranc fracture dislocation, Lisfranc injury, Literature review, Review, Treatment.
INTRODUCTION
The foot is a wonderful organ but frequently an underappreciated part of human body. There are 26 bones, 33 joints, and 107 ligaments in the foot. The number of cycles of loading the foot undergoes in an average person’s life is enough to take them around the world four times. The forces acting on the foot are around 1.5 times the body weight while walking and 3–4 times the body weight while running.1 The functions of the foot include efficient transmission and distribution of weight, providing balance and enhancing the efficiency of locomotion having a great balance of strength, stiffness, and flexibility. And it is able to maintain this efficiency over millions of cycles, unlike any machine. Among the various components of the foot the tarsometatarsal joint also called the Lisfranc joint is a very important component.
BACKGROUND
Lisfranc joint is the articulation between midfoot and forefoot which is composed of the five tarsometatarsal joints. Jacques Lisfranc de Saint–Martin (1790–1847) was a famous French field surgeon in Napoleon’s army who served on the Russian front. In 1815, he developed a new amputation technique at the tarsometatarsal joint level without any osteotomy which enabled quicker procedures for forefoot gangrene for frostbite victims.2 Since then the tarsometatarsal joint has been known as the Lisfranc joint, and the injuries around it are broadly termed as Lisfranc injury: which refers to osseous or ligamentous disruption of the tarsometatarsal and intercuneiform joint complex. There is a spectrum of injuries ranging from stable, ligamentous sprain to grossly displaced and unstable fracture, or fracture dislocation of any of the articular structures of the tarsometatarsal complex. The Lisfranc injuries are rare but if undetected, untreated, or undertreated can cause morbidity and disability.3,4
EPIDEMIOLOGY
The reported incidence of Lisfranc fracture dislocation is 1 per 55,000/year. They constitute approximately 0.2% of all fractures.2 In 20% the diagnosis is late or missed.5 Men are two to four times more likely to sustain these injuries, and commonly in the third decade of life.2 Majority of these (87.5%) are closed injuries6 and more than one third occur in athletes who present with subtle Lisfranc injuries in low-energy sports.7 Myerson et al. found that 58% of the Lisfranc injuries were associated with polytrauma and two thirds of which were due to motor vehicle accidents.3,8
ANATOMY
Osteology
The Lisfranc joint is composed of bony components of medial three tarsometatarsal joints namely the medial, middle, and lateral cuneiforms and first, second, and third metatarsal bases, respectively and the intermetatarsal and anterior intertarsal joints; they are trapezoidal in cross-section with plantar concavity resembling Roman arch also known as the transverse arch. The three medial metatarsal bases articulates with a cuneiform bone, and the lateral two metatarsals articulate with the cuboid.9 The middle cuneiform is recessed compared to the medial and lateral cuneiforms into which the second metatarsal base is positioned making it the apex of the Roman arch in the coronal plane.10 It, therefore, acts as the keystone offering stability to the entire midfoot complex. A shallow second tarsometatarsal joint mortise is considered a risk factor for Lisfranc injury.11
Capsules
There are three separate articular capsules forming three compartments: medial, central, and lateral with separate synovial systems not communicating with each other. The medial compartment encloses the medial cuneiform first metatarsal articulation, the central compartment encloses many joints: the middle cuneiform-second metatarsal joint, lateral cuneiform-third metatarsal joint, intercuneiform joints, second-third intermetatarsal joint, lateral cuneiform-second metatarsal, and lateral cuneiform-fourth metatarsal joints. The lateral compartment encloses the cuboid-fourth and fifth metatarsal joints and the fourth and fifth intermetatarsal joints.12
Ligaments
The Lisfranc ligament complex is composed of dorsal, interosseous, and plantar tarsometatarsal ligaments.
Dorsal Ligaments
These are strong and flat. The first metatarsal is joined to the medial cuneiform by the capsule, and other tarsometatarsal capsules blend with the dorsal and plantar ligaments. The second metatarsal receives a band from each cuneiform, the third from the lateral cuneiform, the fourth from the lateral cuneiform and the cuboid, and the fifth from the cuboid alone.
Plantar Ligaments
These are longitudinal and oblique bands, less regular than the dorsal ligaments. Those for the first and second metatarsals are strongest. The second and third metatarsals are joined by oblique bands to the medial cuneiform and the fourth and fifth metatarsals by a few fibers to the cuboid.
Cuneometatarsal Interosseous Ligaments
There are three of these. One passes from lateral surface of medial cuneiform to the second metatarsal. Known as the Lisfranc’s ligament, it is the strongest and is crucial to the stability of the tarsometatarsal joint complex. Disruption of this ligament can lead to instability and deformity and subsequent degenerative changes. The second connects lateral cuneiform to the second metatarsal. The third connects the lateral cuneiform to the fourth metatarsal base9 (Fig. 1).

Fig. 1: Diagrammatic representation of the Lisfranc ligament complex
Innervation
The interosseous cuneometatarsal ligaments are innervated dorsally by the deep fibular nerve. The plantar aspects of the medial two joints are innervated by the medial plantar nerve and plantar aspects of lateral joints are innervated by lateral plantar nerve.
Factors Maintaining Stability
The major stabilizers are the associated ligaments. The first plantar tarsometatarsal (TMT) ligament has also been shown to be an important stabilizer, especially in the en pointe position, a position commonly used in ballet when the dancer supports the whole of the body weight on the tips of fully extended feet.13 There are no ligaments connecting the first and second metatarsal bases, hence, the risk of divergent displacement during injury. The dorsal ligaments are weaker which explains the occurrence of dorsal displacement.12 The medial interosseous ligament or Lisfranc’s ligament is responsible for the tell-tale avulsion fracture of the second metatarsal base.
Muscles Producing Movement
Movements between the tarsals and metatarsals are limited to flexion and extension, except in the first tarsometatarsal joint, where some abduction and rotation occur caused by tibialis anterior and fibularis longus. Flexion and extension are brought about by the long and short flexors and extensors of the toes.
Columns of the Midfoot
The three columns formed by the osseous and ligamentous complex together the medial, middle, and lateral columns. The medial column refers to the first metatarsal, medial cuneiform, and navicular articulations. The middle column refers to the second and third tarsometatarsal joints and navicular articulations. The fourth and fifth tarsometatarsal joints form the lateral column.9
Biomechanics
The Lisfranc joint is crucial for a normal gait; it is responsible for the equal distribution of weight across the six weight-bearing structures of the forefoot—which are the four metatarsal heads and the two sesamoids plantar to the first metatarsal head. The unique bony arrangement and the ligamentous complex especially the plantar ligament prevent the plantar displacement of the metatarsal bases.
The medial and middle columns have inherently restricted mobility and lateral column has more mobility. There is 10–20° of movement in the lateral column compared to 5–10° of movement in the medial column, the center of mobility in the medial column being at the talonavicular joint whereas the middle column has negligible movement.4 The stiff middle column acts as a rigid lever arm and the relatively stiff medial column and the mobile lateral column provide adjustment for distribution of forces while weight-bearing.
CLASSIFICATION
In 1909, Quenu and Kuss described the classification of Lisfranc injuries based on the three-column concept. The injuries were classified as homolateral, isolated, and divergent.6 This was modified by Hardcastle et al.14 in 1982 into A, B, and C categories. In type A injury all of the metatarsals are displaced in one direction, and there is total incongruity. Type B injury consists of partial incongruity with one or more metatarsals displaced. Finally a type C injury shows a divergent pattern of incongruity.14
A further modified classification system was proposed by Myerson et al., in which the complexity of the injuries were taken into consideration previous type B and type C injuries were subdivided into B1, B2, C1, and C2. In B1 injuries there is isolated medial dislocation of the first metatarsal. In B2 there is isolated lateral dislocation of the second to fifth metatarsals. Type C1 injury is a divergent injury of some of the TMT joints. C2 is a divergent injury of all of the tarsometatarsal joints (Fig. 2).

Fig. 2: X-rays depicting the types of fracture dislocation based on the modified classification of Myerson
MECHANISMS OF INJURY
Lisfranc injuries range from low-energy twisting injuries like trivial sports injury, slip and fall to high-energy injuries like fall from height, motor vehicle accident, high-energy sports and crush injuries. They could be direct or indirect injuries. These injuries happen as a result of combination of axial load and dorsiflexion or plantar flexion, abduction or adduction of the midfoot. The pathoanatomy can vary from pure ligamentous injury, a pure bony injury, or a combination.
Crush injuries can result in open injuries with significant soft tissue compromise especially in the dorsum of foot. First, the dorsal ligaments are disrupted then the stronger plantar Lisfranc ligaments following which bony injury occurs. The bony injury may be at the Lisfranc joint and may extend into the intercuneiform joints, navicular, cuboid proximally, and metatarsal neck and shafts distally. Direct Lisfranc injuries have worst outcomes.
DIAGNOSIS
Details of the exact mechanism of injury should be sought out, which include position of foot at the time of impact, direction of force, and the velocity involved. In low-velocity injuries patient gives history of feeling a pop/give away sensation in the midfoot and painful weight-bearing.
Overlying soft tissue is inspected for abrasions, lacerations, and diffuse swelling. Plantar arch ecchymosis is considered pathognomonic of a Lisfranc injury, but it may be absent in low-energy injuries.15
Tenderness at midfoot and pain on passive movement of the forefoot are suggestive of Lisfranc injury.16 Altered sensation over the dorsum of the first intermetatarsal space due to neuropraxia of the medial terminal branch of the deep peroneal nerve is elicited with decreased two point discrimination.
Provocative maneuvers like passive abduction and pronation of forefoot with the hindfoot fixed elicits pain. The ‘Piano Key Test’ which is performed by anteroposterior (AP) translation of the head of the involved metatarsal after stabilizing the forefoot and hindfoot is useful to isolate the location of the injury. The ‘Positive Gap’ which is the increase in distance between the first two toes correlates with intercuneiform instability.3
Radiographic evaluation is with nonweight-bearing AP, oblique, and lateral views of the foot. But since 20% of unstable Lisfranc injuries are missed in plain radiographs, stress views are helpful in low-energy injuries,17 and compared with the normal side.18 On weight-bearing AP view of the foot, the medial and lateral borders of the first metatarsal should be aligned with medial cuneiform; the medial border of the second metatarsal should align with the medial border of the middle cuneiform. The ‘fleck sign’ is the presence of a small bone fragment in the first intermetatarsal space indicating the avulsion of Lisfranc ligament (Fig. 3). In the weight-bearing internal oblique view, the medial border of the third metatarsal should be aligned with the medial border of the lateral cuneiform; the medial border of the fourth metatarsal should align with the medial border of the cuboid. On weight-bearing lateral view, the medial and middle columns should symmetrically align with the long axis of talus. Disruption of any of these alignments is indicative of Lisfranc injury.3 Any dorsal displacement of the metatarsal base with respect to cuneiform is abnormal (Fig. 4).
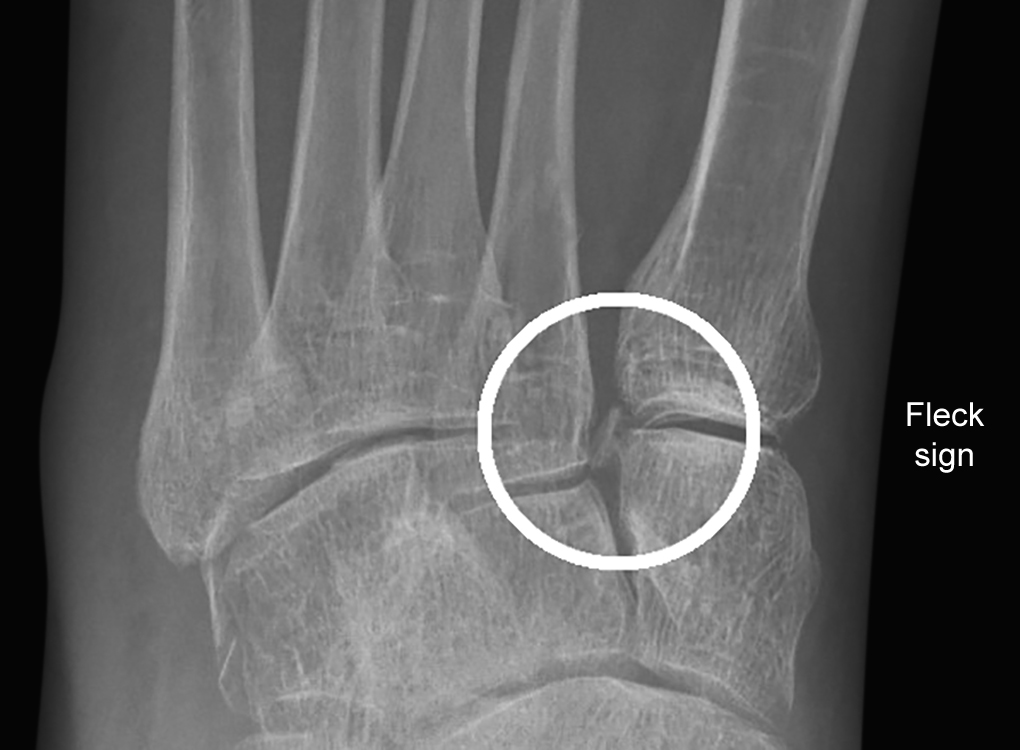
Fig. 3: Fleck sign representing avulsion of Lisfranc ligament
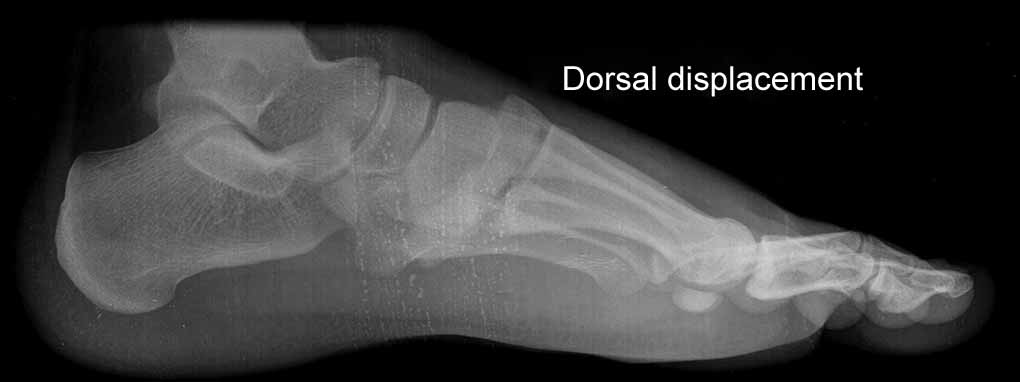
Fig. 4: Dorsal displacement of metatarsal base in relation to cuneiform
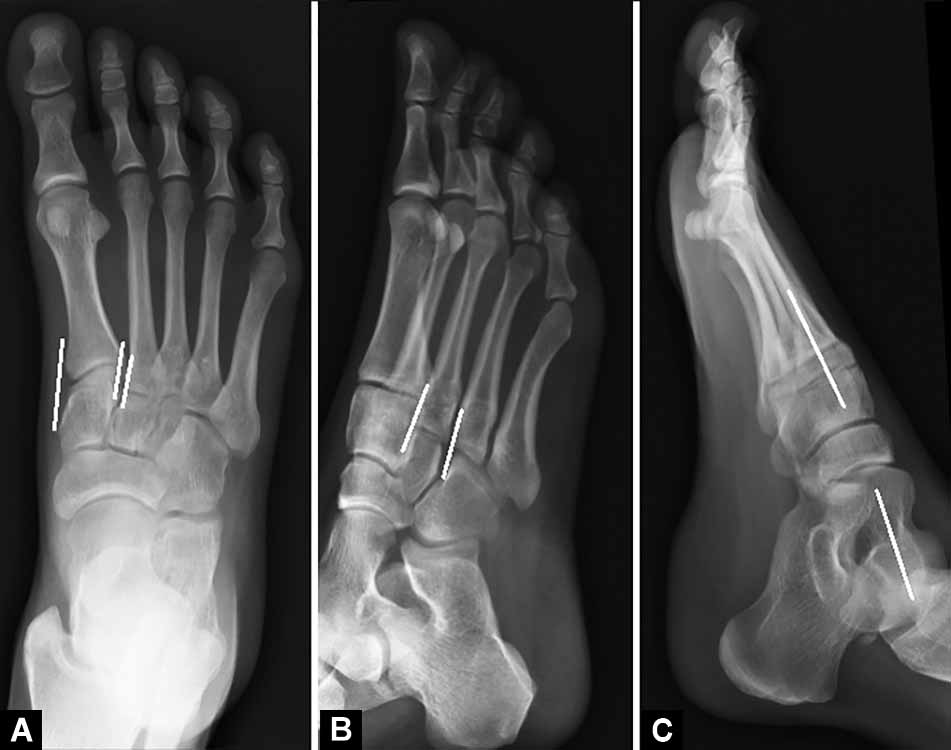
Figs 5A to C: Normal radiological parameters. (A) AP view of the foot, the medial and lateral borders of the first metatarsal should be aligned with medial cuneiform; the medial border of the second metatarsal should align with the medial border of the middle cuneiform. (B) Oblique view, the medial border of the third metatarsal should be aligned with the medial border of the lateral cuneiform; the medial border of the fourth metatarsal should align with the medial border of the cuboid. (C) Lateral view, the medial and middle columns should symmetrically align with the long axis of talus
Associated injuries include “nut cracker” fractures of the cuboid—which occurs due to compression of cuboid by fourth and fifth metatarsal bases.19-23
Ultrasonography has been described recently as diagnostic tool for evaluating Lisfranc injuries.24 The integrity of the dorsal ligament and the distance between the medial cuneiform and second metatarsal base are assessed. However, its accuracy for diagnosing Lisfranc instability is yet to be definitively proven.24
Computed tomography scans are beneficial in subtle injuries, especially in polytrauma and in patients with other injuries which prevent weight-bearing. Other occult fractures extending into the forefoot and hindfoot can be detected. Computed tomography scan is useful for preoperative planning.25 Computed tomography has several advantages over radiography since they are free of overlapping, superimposed structures, and are able to detect occult fractures, differentiate small avulsion fractures (fleck sign) from its mimics (i.e., os intermetatarseum which are located more dorsally and have a more rounded shape), clarify the degree of comminution with or without an intra-articular extension, and demonstrate minimal osseous subluxation around the TMT joints.26 Preidler et al. showed that CT revealed 60% more metatarsal fractures and twice as many tarsal fractures and joint malalignments as radiography.27 Sripanich et al. strongly recommend CT for patients with high-energy midfoot injuries or complex fractures.18
Magnetic resonance imaging is useful in subtle injuries to detect ligamentous injuries without bony involvement.20 It has a sensitivity and predictive value of up to 94% in determining instability of the Lisfranc joint (Fig. 5).28
Occasionally both CT and MRI may be needed to fully assess the injuries. However, intraoperative assessment29 with direct visualization is still the best definitive diagnostic tool to evaluate the degree of Lisfranc instability and TMT malalignment with close to 100% accuracy.18
NATURAL HISTORY
If unstable injuries are missed the natural history of Lisfranc fracture dislocation is development of malreduced and malaligned tarsometatarsal joints with persistent midfoot pain, activity limitations, nonunion of fracture, and posttraumatic arthritis eventually requiring arthrodesis.30 The collapse of the keystone region subsequently results in pes planusdeformity. Functionally patient complaints of deformed foot, painful weight-bearing, and stiffness.
DEFINING STABILITY
The definition of instability presently is greater than 2 mm of residual tarsometatarsal displacement or more than 15° of persistent talo-first metatarsal angulation. Injuries that present with painful weight-bearing, pain with metatarsal motion, and tenderness to palpation but fail to exhibit any instability on stress radiographs should be considered a sprain.31 Curtis et al. classified the tarsometatarsal injuries into first degree, second degree, and third degree sprains (with diastasis between the metatarsals or cuneiforms), fractures, and fracture dislocations. They found that third degree sprains, fractures, and fracture dislocations require anatomical reduction and internal fixation for optimal functional results.32 So first and second degree sprains can be considered stable injuries and third degree sprains and beyond can be considered unstable injuries.
TREATMENT
Closed Injuries
Reduction Techniques
Anatomical reduction and stable internal fixation is the standard of care for treating unstable Lisfranc injuries.33 The method of reduction is debated, while some authors believe closed reduction and percutaneous fixation is the preferred technique others believe that open reduction and internal fixation is the appropriate method.
Closed reduction
The closed reduction is generally achieved by manipulative maneuvers which start with manual stabilization of the hindfoot followed by axial traction of the forefoot with manual compression of the second metatarsal base. Once the second metatarsal is located into the mortise the first metatarsal is reduced by giving axial traction to big toe with varus and thrust on the metatarsal base from dorsal downward and from medial to lateral. If there is residual diastasis then stab incisions are made over the dorsum of second metatarsal and another over the medial surface of the medial cuneiform followed by placing reduction clamps through the stab incisions and final anatomical reduction is achieved by compressing the medial cuneiform toward the second metatarsal base. This maneuver often realigns the entire articulation because the second metatarsal pulls the third and fourth metatarsals with it. If the lateral column is unstable, it is reduced with axial traction and direct pressure over the metatarsal base.
Literature supports that anatomic alignment is necessary, and good or excellent results have been achieved in 50–95% of patients with anatomic alignment, compared with 17–30% of patients with nonanatomic alignment.34 Hence, if anatomical reduction cannot be achieved with closed reduction then it is mandatory to proceed to open reduction.
Open reduction
Malreduction is usually due to interposition of a fracture fragment or soft tissue at the base of the second metatarsal, preventing anatomical apposition between the medial and middle columns. In such instances an open reduction is performed, the small fragment of bone can be pushed plantar to the first metatarsal to achieve reduction. Sometimes soft tissue may be interposed and dislocation of the tendon of tibialis anterior between the middle and medial columns into the medial tarsal metatarsal joint has been reported.
A longitudinal incision is made over the dorsomedial aspect between the first and second tarsometatarsal joint. The medial branch of the superficial peroneal nerve is identified and protected. Surgical approach is in the interval between the extensor hallucis longus and the anterior tibialis tendons which are retracted to the sides and protected. The dorsalis pedis artery and deep peroneal nerve which are just lateral to the extensor hallucis longus (EHL) tendon should also be protected. If the adjacent lateral metatarsals do not reduce, an incision is made between the fourth and fifth metatarsals. This must be more lateral for a crush type fracture of the cuboid. To restore the length of the cuboid, bone grafting has been described by some authors.35 Marín-Peña et al. reported no statistically significant difference between overall American Orthopaedic Foot and Ankle Society (AOFAS) in open reduction K-wire fixation and closed reduction K-wire fixation groups and that there was a poor association between the extent of radiological arthritis and clinical scores.36
FIXATION MODALITIES
K-wires
Once anatomical reduction is achieved 1.8 mm K-wire is passed from the metaphyseal region of the first metatarsal into the medial cuneiform through stab incisions. Following this the second metatarsal is stabilized similarly by passing the K-wire into the middle cuneiform. At least two K-wires in crossed fashion are necessary for adequate stability for each tarsometatarsal joint and they should be inserted up to navicular at least for optimal stability, they can be inserted into talus also if required. Joint stiffness is usually not a problem even if these joints are temporarily crossed by K-wires. This maneuver reduces the rest of the articulations of the lateral column along with the middle column toward the middle cuneiform obliquely. The lateral column is also stabilized in a similar fashion with K-wires through each metatarsal into the cuboid. Intermetatarsal transverse nonparallel K-wire configuration can also be used.37
Infection is always a possibility when pins are left outside skin, and the reduction will be compromised if they have to be removed prematurely. Therefore, if pins are used, they should be buried subcutaneously for later retrieval, which would have to be done under anesthesia, however, if they are left prominent outside removal is much economical.35
K-wire fixation is advantageous since it avoids further soft tissue devitalization and produces further minimal articular cartilage damage. The ability to insert K-wires away from compromised soft tissues and that they can be removed during outpatient visits are added advantages. In our previous series of patients with open Lisfranc injuries there was no secondary loss of reduction while using only K-wire fixation for these injuries show evidence of outcome with K-wire fixation.37
Screws
The sequence of fixation of screws are similar to that described for K-wires, generally 3.5 mm cortical screws are used to transfix the metatarsal base to the cuneiforms. Countersinking the screw head is essential to prevent fracture of the proximal metatarsal. If a dislocation of the middle column is present, it is preferable to use a larger, partially threaded 4.5 mm cancellous screw to reduce and hold the second metatarsal in its mortise; this is introduced from the medial cuneiform into the second metatarsal. The entry point for the screw into the second metatarsal should be approximately 16–20 mm distal to the joint and the screw should be introduced at an angle of 30° to the metatarsal shaft. The lateral column screw fixation is debated since it increases the stiffness.35
The advantage of screw fixation is that it is more rigid compared to K-wires, however, the drawback is the surface area breached by the screws in the articular cartilage is much more than that of K-wires. Very often multiple drilling attempts are made for accurate screw placement which further increases articular cartilage damage. The other disadvantages are that these screws can cause soft tissue impingement and would otherwise also require a secondary procedure for removal after 6 months, potential screw breakage, articular damage to the involved joints, and the risk of subsequent osteoarthritis. In 16.1% patients it was necessary to remove broken or problem-causing screws. Some reports have described the use of absorbable screws in order to overcome these complications. However, it is too early to draw any conclusions about this method.38 Most authors prefer screw fixation of the medial and middle columns and K-wire fixation of the lateral column.39
Plate Fixation
Incisions are made as described for open reduction. The periosteum of first TMT joint is incised longitudinally to expose the dislocation, as well as the second TMT joint and the intercuneiform articulation. A thorough debridement is performed to remove any interposed tissue within the joints. Temporary K-wire fixation may be required before plate fixation when the fracture reduction is difficult to maintain. Mini plates like 2.7 mm “L” and “T” plates are used. The distal portion of plate is fixed with screws onto the first metatarsal, and the proximal portion of plate is fixed to medial cuneiform. Care is taken not to place the plates in the course of extensor tendons especially under the extensor hallucis. The second and third tarsometatarsal joint is also fixed similarly. The second incision may be centered between the fourth and fifth metatarsals, and the fourth or fifth tarsometatarsal joint can be fixed through this incision by plate.
Plate fixation is described as an alternate method to maintain reduction; they, however, can be performed only when soft tissue conditions are good.40 Compared to transarticular fixations like K-wires and screw fixations plate fixation helps in preventing iatrogenic articular cartilage damage. Plates also have other advantages when compared to transarticular screws; if the screws break, distal threads are typically intra-articular and may contribute to further articular cartilage damage with movement and they are difficult to remove without additional significant damage. Whereas if a plate breaks in the joint, joint movement can still occur and it may not be necessary to remove it. If the screws inserted through the plate are broken, the distal threads would be left in the metatarsals or cuneiforms with no risk of more irritation of soft tissues or articular cartilage; or else they are easily accessible without contacting the joint, by specific instruments to remove screws. Vecchio et al. claim that the morbidity risk would be lower in plate fixation and weight-bearing can be achieved early on after surgery leading to less joint rigidity, muscle atrophy, and disuse osteopenia.41
The disadvantage of plates as compared to screws is that plates cannot be inserted percutaneously, and so an open reduction is required in patients who might simply need a closed reduction. Several incisions are typically needed, which increases the risk of skin necrosis and delayed wound healing.42 Plates are more likely to cause soft tissue irritation due to their prominence: many surgeons remove the implant routinely 3–4 months after the fixation.42
In a cadaveric study done by Alberta et al. there was no significant difference noted with direct comparison between plates and screws with respect to ability to realign the first and second and to maintain TMT joint alignment during loading. The amount of articular surface destruction caused by one 3.5-mm screw was 2.0 ± 0.7% for the medial cuneiform, 2.6 ± 0.5% for the first metatarsal, 3.6 ± 1.2% for the intermediate cuneiform, and 3.6 ± 1.0% for the second metatarsal.43 Hu et al. reported a mean AOFAS midfoot score of 83.1 points in the plate fixation group and 78.5 points in the screw fixation group.40 Lau et al. in their study comparing the functional outcomes after transarticular screws, dorsal bridge plating or combination treatment concluded that outcomes are most dependent on the quality of anatomical reduction and not the choice of fixation implant used,44 whereas Kirzner et al. found that there is significantly better functional and radiological outcome following dorsal plating. Primary arthrodesis may be a preferred option for more severe injuries such as those of Myerson types A and C2.8,45
Primary Arthrodesis
Primary arthrodesis may be indicated in comminuted fractures; since stiffness is preferred over instability to maintain the rigidity of the medial and middle columns during gait and in case with severe cartilage damage, preexisting midfoot arthritis, and delayed diagnosis as well as in patients with neuropathic injuries.34
Incisions are made as described for open reduction. Articular cartilage and fibrous tissue are removed from the affected tarsometatarsal joints. Fixation for the medial cuneiform bone to the base of second metatarsal and first to third tarsometatarsal joints are done preferably with 4.0 cannulated screws in a similar manner to open reduction and internal fixation (ORIF), the only difference here being the joint surfaces are compressed against each other to achieve fusion.
Studies which compared ORIF with primary arthrodesis report that patients treated with primary arthrodesis had a quicker recovery, a higher foot function index score, and a superior return to function and the reoperation rate was significantly lower than those who underwent ORIF.46 Barnds et al. reported that primary arthrodesis is both significantly more expensive and has a higher complication rate than ORIF.47
Postoperative rehabilitation is the same in all groups and consisted of 6 weeks nonweight-bearing, followed by protected weight-bearing with full ankle range of motion until 3 months, after which full weight-bearing is allowed gradually based on follow-up assessment.
Newer Techniques
Recently various new techniques are being described for ligament reconstruction in order to achieve flexible tarsometatarsal joints as an end result.48 Minimally invasive medial plating for low-energy Lisfranc injuries has also been attempted.41 These techniques, however, lack long-term follow-up on functional outcome.
OPEN AND CRUSH INJURIES
Any open injuries or dislocations causing soft tissue tension should be reduced and stabilized and temporarily immobilized with plaster or external fixation and followed by definitive fixation 1 or 2 weeks later once soft tissue conditions improve.49 Compartment syndrome is also a possibility in crush injuries which should be dealt judiciously with external fixation and fasciotomies as required.
In open Lisfranc injuries the treatment protocol differs primarily with the choice of fixation. Fixation with K-wires is preferred over internal fixation with screw and plates in view of the poor soft tissue conditions. Following thorough debridement anatomical reduction is achieved followed by passing of 1.8 mm K-wires in a retrograde fashion through each metatarsal medullary canal onto the cuneiforms, and can be extended up to the navicular in unstable situations or augmented with external fixation followed by secondary soft tissue cover.37 In open injuries the exposed cancellous bony surface enables good new bone formation, so by maintaining the midfoot arch and tarsometatarsal alignment sound fusion can be achieved (Fig. 6).

Fig. 6: Midfoot fusion in an open Lisfranc injury
COMPLICATIONS
The most common short-term postoperative complications include compartment syndrome, deep vein thrombosis, and infection, which can be clinically suspected. Imaging may be more helpful for detecting long-term complications, including midfoot osteoarthritis, planovalgus deformity, and hardware failure (i.e., screw loosening, migration, or breakage).39 Stavlas et al. in their systematic review reported compartment syndrome in 2.6%, skin problems in 3.6%, infection in 1.5%, deep vein thrombosis in 0.5%, reflex sympathetic dystrophy in 1.0%, screw problems in 16.1%, and amputation in 1.0% of the patients. Posttraumatic arthritis on radiographic examination was reported in 49.6%.6
Osteoarthritis is a well-recognized complication of Lisfranc injuries, with about 50% of patients showing radiographic signs of Lisfranc osteoarthritis after ORIF, of these only 8% patients become symptomatic enough to require arthrodesis. One of the major determinants to the development of arthritis is restoration and maintenance of anatomic alignment.6 Secondary arthrodesis remains an option even after the failure of other treatment modalities.50
CONCLUSION
Though Lisfranc injury is rare it causes significant disabilities if not treated well. It can be caused by both low- and high-energy injuries. Careful history taking, clinical examination, and appropriate radiographic imaging are helpful in the diagnosis. In high-energy unstable injuries normal radiographic parameters are not preserved. In low-energy unstable injuries where clinical suspicion is high stress radiographs are indicated, CT and MRI are of additional help for confirming diagnosis and preoperative planning.
In displaced unstable Lisfranc injuries anatomical reduction and stable internal fixation is the standard of care. The common complications include osteoarthritis, persistent pain and gait abnormalities, implant related problems, and skin problems. There are many modalities of fixation available, however, final outcome depends on early diagnosis, achieving anatomic reduction and maintaining it, irrespective of the method of reduction or the mode of fixation.
ORCID
Jeremy Bliss https://orcid.org/0000-0003-3973-5058
Sandeep Albert https://orcid.org/0000-0002-4427-3460
REFERENCES
1. Nilsson J, Thorstensson A. Ground reaction forces at different speeds of human walking and running. Acta Physiol Scand 1989;136(2):217–227. DOI: 10.1111/j.1748-1716.1989.tb08655.x
2. Desmond EA, Chou LB. Current concepts review: Lisfranc injuries. Foot Ankle Int 2006;27(8):653–660. DOI: 10.1177/107110070602700819
3. Moracia-Ochagavía I, Rodríguez-Merchán EC. Lisfranc fracture-dislocations: current management. EFORT Open Rev 2019;4(7):430–444. DOI: 10.1302/2058-5241.4.180076
4. Coetzee JC. Making sense of Lisfranc injuries. Foot Ankle Clin 2008;13(4):695–704, ix. DOI: 10.1016/j.fcl.2008.07.001
5. Rammelt S, Schneiders W, Schikore H, et al. Primary open reduction and fixation compared with delayed corrective arthrodesis in the treatment of tarsometatarsal (Lisfranc) fracture dislocation. J Bone Joint Surg Br 2008;90(11):1499–1506. DOI: 10.1302/0301-620X.90B11.20695
6. Stavlas P, Roberts CS, Xypnitos FN, et al. The role of reduction and internal fixation of Lisfranc fracture–dislocations: a systematic review of the literature. Int Orthop 2010;34(8):1083–1091. DOI: 10.1007/s00264-010-1101-x
7. Benirschke SK, Meinberg E, Anderson SA, et al. Fractures and dislocations of the midfoot: Lisfranc and Chopart injuries. J Bone Joint Surg Am 2012;94(14):1325–1337. DOI: 10.2106/jbjs.l00413
8. Myerson MS, Fisher RT, Burgess AR, et al. Fracture dislocations of the tarsometatarsal joints: end results correlated with pathology and treatment. Foot Ankle 1986;6(5):225–242. DOI: 10.1177/107110078600600504
9. Standring S. Gray’s Anatomy: The Anatomical Basis of Clinical Practice. 41st ed.; 2020. Chapter 84, Ankle and foot: tarsometatarsal joints section.
10. Scolaro J, Ahn J, Mehta S. Lisfranc fracture dislocations. Clin Orthop 2011;469(7):2078–2080. DOI: 10.1007/s11999-010-1586-z
11. Peicha G, Labovitz J, Seibert FJ, et al. The anatomy of the joint as a risk factor for Lisfranc dislocation and fracture-dislocation. An anatomical and radiological case control study. J Bone Joint Surg Br 2002;84(7):981–985. DOI: 10.1302/0301-620x.84b7.12587
12. Castro M, Melão L, Canella C, et al. Lisfranc joint ligamentous complex: MRI with anatomic correlation in cadavers. Am J Roentgenol 2010;195(6):W447–W455. DOI: 10.2214/AJR.10.4674
13. Kadel N, Boenisch M, Teitz C, et al. Stability of Lisfranc joints in ballet pointe position. Foot Ankle Int 2005;26(5):394–400. DOI: 10.1177/107110070502600510
14. Hardcastle PH, Reschauer R, Kutscha-Lissberg E, et al. Injuries to the tarsometatarsal joint. Incidence, classification and treatment. J Bone Joint Surg Br 1982;64(3):349–356. DOI: 10.1302/0301-620X.64B3.7096403
15. Ross G, Cronin R, Hauzenblas J, et al. Plantar ecchymosis sign: a clinical aid to diagnosis of occult Lisfranc tarsometatarsal injuries. J Orthop Trauma 1996;10(2):119–122. DOI: 10.1097/00005131-199602000-00008
16. Aronow MS. Treatment of the missed Lisfranc injury. Foot Ankle Clin 2006;11(1):127–142, ix. DOI: 10.1016/j.fcl.2005.12.005
17. Goossens M, De Stoop N. Lisfranc’s fracture-dislocations: etiology, radiology, and results of treatment. A review of 20 cases Clin Orthop 1983;(176):154–162. PMID: 6851319.
18. Sripanich Y, Weinberg MW, Krähenbühl N, et al. Imaging in Lisfranc injury: a systematic literature review. Skeletal Radiol 2020;49(1):31–53. DOI: 10.1007/s00256-019-03282-1
19. Gallagher SM, Rodriguez NA, Andersen CR, et al. Anatomic predisposition to ligamentous Lisfranc injury: a matched case-control study. J Bone Joint Surg Am 2013;95(22):2043–2047. DOI: 10.2106/JBJS.K.01142
20. Preidler KW, Brossmann J, Daenen B, et al. MR imaging of the tarsometatarsal joint: analysis of injuries in 11 patients. AJR Am J Roentgenol 1996;167(5):1217–1222. DOI: 10.2214/ajr.167.5.8911184
21. Kitsukawa K, Hirano T, Niki H, et al. MR imaging evaluation of the Lisfranc ligament in cadaveric feet and patients with acute to chronic Lisfranc injury. Foot Ankle Int 2015;36(12):1483–1492. DOI: 10.1177/1071100715596746
22. Shapiro MS, Wascher DC, Finerman GA. Rupture of Lisfranc’s ligament in athletes. Am J Sports Med 1994;22(5):687–691. DOI: 10.1177/036354659402200518
23. Lau S, Bozin M, Thillainadesan T. Lisfranc fracture dislocation: a review of a commonly missed injury of the midfoot. Emerg Med J 2016;34(1):52–56. DOI: 10.1136/emermed-2015-205317
24. DeLuca MK, Walrod B, Boucher LC. Ultrasound as a diagnostic tool in the assessment of Lisfranc joint injuries. J Ultrasound Med 2020;39(3):579–587. DOI: 10.1002/jum.15138
25. Clare MP. Lisfranc injuries. Curr Rev Musculoskelet Med 2017;10(1):81–85. DOI: 10.1007/s12178-017-9387-6
26. Chan BY, Markhardt BK, Williams KL, et al. Os conundrum: identifying symptomatic sesamoids and accessory ossicles of the foot. AJR Am J Roentgenol 2019;213(2):417–426. DOI: 10.2214/AJR.18.20761
27. Preidler KW, Peicha G, Lajtai G, et al. Conventional radiography, CT, and MR imaging in patients with hyperflexion injuries of the foot: diagnostic accuracy in the detection of bony and ligamentous changes. AJR Am J Roentgenol 1999;173(6):1673–1677. DOI: 10.2214/ajr.173.6.10584818
28. Raikin SM, Elias I, Dheer S, et al. Prediction of midfoot instability in the subtle Lisfranc injury. Comparison of magnetic resonance imaging with intraoperative findings. J Bone Joint Surg Am 2009;91(4):892–899. DOI: 10.2106/jbjs.h.01075
29. Seo DK, Lee HS, Lee KW, et al. Nonweightbearing radiographs in patients with a subtle Lisfranc injury. Foot Ankle Int 2017;38(10):1120–1125. DOI: 10.1177/1071100717717220
30. Komenda GA, Myerson MS, Biddinger KR. Results of arthrodesis of the tarsometatarsal joints after traumatic injury. J Bone Joint Surg Am 1996;78(11):1665–1676. DOI: 10.2106/00004623-199611000-00005
31. Reid JJ, John S. Rockwood and Green’s Fractures in Adults. 7th ed. Lippincott Williams & Wilkins; 2010. Early fractures and dislocations of the midfoot and forefoot; p. 2129–2140.
32. Curtis MJ, Myerson M, Szura B. Tarsometatarsal joint injuries in the athlete. Am J Sports Med 1993;21(4):497–502. DOI: 10.1177/036354659302100403
33. Perugia D, Basile A, Battaglia A, et al. Fracture dislocations of Lisfranc’s joint treated with closed reduction and percutaneous fixation. Int Orthop 2003;27(1):30–35. DOI: 10.1007/s00264-002-0396-7
34. Watson TS. Treatment of Lisfranc joint injury: current concepts. J Am Acad Orthop Surg 2010;18(12):718–728. DOI: 10.5435/00124635-201012000-00002
35. Myerson MS. The diagnosis and treatment of injury to the tarsometatarsal joint complex. J Bone Joint Surg Br 1999;81(5):756–763. DOI: 10.1302/0301-620x.81b5.10369
36. Marín-Peña OR, Recio FV, Gómez TS, et al. Fourteen years follow up after Lisfranc fracture-dislocation: functional and radiological results. Injury 2012;43(Suppl 2):S79–S82. DOI: 10.1016/S0020-1383(13)70185-2
37. Nithyananth M, Boopalan PRJVC, Titus VTK, et al. Long-term outcome of high-energy open Lisfranc injuries: a retrospective study. J Trauma Inj Infect Crit Care 2011;70(3):710–716. DOI: 10.1097/TA.0b013e3181f02ab9
38. Thordarson DB, Hurvitz G. PLA screw fixation of Lisfranc injuries. Foot Ankle Int 2002;23(11):1003–1007. DOI: 10.1177/107110070202301106
39. Siddiqui NA, Galizia MS, Almusa E, et al. Evaluation of the tarsometatarsal joint using conventional radiography, CT, and MR imaging. Radiographics 2014;34(2):514–531. DOI: 10.1148/rg.342125215
40. Hu SJ, Chang SM, Li XH, et al. Outcome comparison of Lisfranc injuries treated through dorsal plate fixation versus screw fixation. Acta Ortop Bras 2014;22(6):315–320. DOI: 10.1590/1413-78522014220600576
41. Vecchio D, Javier J, Ghioldi M, et al. minimally invasive medial plating of low-energy Lisfranc injuries: preliminary experience with five cases. Adv Orthop 2016;2016:4861260. DOI: 10.1155/2016/4861260
42. Wagner E, Ortiz C, Villalón IE, et al. Early weight-bearing after percutaneous reduction and screw fixation for low-energy Lisfranc injury. Foot Ankle Int 2013;34(7):978–983. DOI: 10.1177/1071100713477403
43. Alberta FG, Aronow MS, Barrero M, et al. Ligamentous Lisfranc joint injuries: a biomechanical comparison of dorsal plate and transarticular screw fixation. Foot Ankle Int 2005;26(6):462–473. DOI: 10.1177/107110070502600607
44. Lau S, Howells N, Millar M, et al. Plates, screws, or combination? Radiologic outcomes after Lisfranc fracture dislocation. J Foot Ankle Surg 2016;55(4):799–802. DOI: 10.1053/j.jfas.2016.03.002
45. Kirzner N, Zotov P, Goldbloom D, et al. Dorsal bridge plating or transarticular screws for Lisfranc fracture dislocations. Bone Jt J 2018;100-B(4):468–474. DOI: 10.1302/0301-620X.100B4.BJJ-2017-0899.R2n>
46. Henning JA, Jones CB, Sietsema DL, et al. Open reduction internal fixation versus primary arthrodesis for Lisfranc injuries: a prospective randomized study. Foot Ankle Int 2009;30(10):913–922. DOI: 10.3113/fai.2009.0913
47. Barnds B, Tucker W, Morris B, et al. Cost comparison and complication rate of Lisfranc injuries treated with open reduction internal fixation versus primary arthrodesis. Injury 2018;49(12):2318–2321. DOI: 10.1016/j.injury.2018.10.002
48. Nery C, Giza E, Wagner E, et al. Dynamic Lisfranc joint repair concept: surgical technique for a synthetic neoligamentplasty. Muscle Ligaments Tendons J 2019;9(4):562–570. DOI: 10.32098/mltj.04.2019.11
49. Cassinelli SJ, Moss LK, Lee DC, et al. Delayed open reduction internal fixation of missed, low-energy Lisfranc injuries. Foot Ankle Int 2016;37(10):1084–1090. DOI: 10.1177/1071100716655355
50. Mulier T, Reynders P, Sioen W, et al. The treatment of Lisfranc injuries. Acta Orthop Belg 1997;63(2):82–90. PMID: 9265792.
________________________
© The Author(s). 2023 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.